Conduction System Pacing with Dr Bill Petrellis

Specialising in general adult cardiology and electrophysiology, including atrial fibrillation and device implantation
Clinic locations: Bella Vista and Chatswood
Progressive Evolution of Pacing Technology
Admittedly, there has been little to be excited about regarding cardiac pacing over the years. Pacemakers are commonplace, lifesaving implantable devices that have been available for decades allowing us to manage patients with symptomatic bradycardia, whether due to slow atrial fibrillation, sinus node dysfunction or heart block. There has been a progressive evolution of pacing technology over the years. Devices are smaller, battery longevity has increased and pacing algorithms have been developed to manage atrial arrhythmias and to reduce the burden of unnecessary ventricular pacing. Leadless pacemakers have recently been developed, which pace the heart via an all-in-one battery and electrode “capsule” delivered to and anchored within the right ventricular chamber.
Future improvements will include “far field” atrial sensing of a single chamber device and the use of two “capsules”, one in the atrium and another in the ventricle, connected wirelessly to permit AV synchrony, an important factor which helps preserve cardiac efficiency. Traditional pacing techniques involve placing a lead within the RV, usually at the apex and more recently, the mid RV septum. Pacing the right ventricle alone induces electrical dyssynchrony, that is, the right ventricle contracts first and left ventricle is activated much later as the action potential propagates by slow conduction from the right ventricle via the myocardium, across the interventricular septum, and then on to activate the left ventricle. The electrical representation of this phenomenon is the broad, abnormal appearance of the paced ventricular QRS complex, which is effectively, pacing induced left bundle branch block (LBBB). The mechanical consequence of ventricular dyssynchrony is its potential to induce heart failure, a condition commonly referred to as pacemaker mediated cardiomyopathy. Other detrimental effects include an increased risk of atrial fibrillation and increased mortality. As such, there has been much effort in the study of alternate site pacing.
Cardiac Resynchronisation Therapy (CRT)
The advent of CRT at the turn of the century, was a major advancement in the management of heart failure patients with intrinsic LBBB. This technique involves positioning one lead within the right ventricle (RV) and another to pace the left ventricle (LV) via a lead advanced into a suitable coronary sinus tributary overlying the lateral left ventricular myocardium. The timing of left and right ventricular pacing can be adjusted to reduce dyssynchrony caused by the pre-existing LBBB. Most commonly, the LV lead is activated before the RV lead by a mere 20 to 40 milliseconds resulting in a narrower paced QRS pattern. The seemingly insignificant advancement of left ventricular activation results in a marked improvement of LV systolic contraction and reduced functional mitral regurgitation in up to 70% of patients. Occasionally, cardiac resynchronisation results in normalisation of the ejection fraction, and these individuals are known as super responders. However, CRT is limited by the coronary sinus anatomy, the presence of LV scar and the inherent potential for phrenic nerve stimulation, a structure which overlies the lateral left ventricle.
Placing an LV lead is often technically challenging, not always successful, and furthermore, 30% of recipients are non-responders despite successful LV lead placement. I recall during the second year of my advanced cardiology training at the Austin Hospital in 2002, a newly minted consultant cardiologist, who had just returned from the United States, requested CRT for an inpatient with heart failure. The experienced senior staff electrophysiologist accepted the challenge to implant his first CRT device. I recall his frustration while we spent several hours at the private hospital on a Saturday morning, trying to position a simple bipolar lead in the coronary sinus using rudimentary sheaths. Today, we have an almost endless catalogue of sheaths, additional tools and quadripolar leads, which have significantly increased the success rate of lead placement and improved RV/LV resynchronisation. Multipolar pacing and AV synchrony algorithms have further added to cardiac mechanical optimisation and perhaps CRT response.
Conduction system pacing is a technique that recruits the native His-Purkinje system producing physiological pacing which results in a narrow paced ventricular QRS complex. It therefore avoids the aforementioned phenomenon of electrical dyssynchrony induced by standard pacing techniques, including CRT. Fast forward to 2004 during the second year of my Electrophysiology and Pacing fellowship at the University of Western Ontario in London, Canada. I quickly became a proficient implanter, spending a day or two each week grinding through my list of 5-7 new implants, upgrades, and generator replacements. But there was one thing I really didn’t look forward to - my scheduled afternoon in the pacing research lab. One of my supervising professors in London was researching the viability of His bundle pacing and comparing its haemodynamics (including cardiac output) to conventional RV apical pacing. The His bundle is a collection of heart muscle cells specialised for electrical conduction. It transmits electrical impulses from the atrioventricular node (AVN) to the bundle branches, causing the ventricles to contract. Hours were spent testing His bundle pacing on consenting pacemaker recipients, positioning, and fixing the lead, pacing at different rates and measuring heamodynamics, only to remove it and leave the final permanent lead at the RV apex. At the time, I did not appreciate the significance of his research, nor did I find it remotely interesting, for shame…. then there was radio silence for over a decade.
Conduction System Pacing
An enthusiastic explosion for conduction system pacing occurred around 2015, which was building over the preceding 5 years. #don’t-dis-the His (https://twitter.com/hashtag/ dontdisthehis?lang=en) was its catch cry. Imagine, cardiac pacing which produces a normal paced QRS complex and the ability to correct intrinsic LBBB. Yes, you read correctly, pacing the His bundle above the site of LBBB often corrected it to produce a narrow paced QRS. Fantastic! I was invited to attend a His bundle pacing course at Geelong in July 2017.
We observed several cases and attended lectures outlining the technique. I was excited to return to Sydney and offer it to my patients, but my initial enthusiasm quickly waned. I was discouraged that the “landing site” for the His bundle lead was rather small making it a tricky implant. I also noted that the lead dislodgement rate was much higher than conventional pacing, requiring lead repositioning with its inherent high risk of infection, not to mention patient inconvenience. More importantly was the observation of increasing His capture threshold over time and the potential for non-capture, an unacceptable outcome for pace dependant patients with complete heart block. Another frequent observation was that loss of His bundle capture resulted in capture of the septal myocardium, so the benefi t of physiological pacing was lost, despite all efforts.
Pacemaker follow up is also time consuming as there is a necessity to confi rm His bundle capture using a 12 lead ECG, not to mention issues arising from lead over- and under sensing. My conclusion was that although His bundle pacing is the most elegant solution in theory, it is not so in practice. In November 2019, I had the opportunity to visit Fuwai Hospital, National Cardiovascular Centre, in Beijing China. This impressive true “centre of excellence” is focussed solely on the management of cardiovascular disease. The facility boasts 700 beds, 12 operating rooms, 2 hybrid operating rooms and 10 interventional catheterization rooms. On offer was an excellent training program for left bundle area pacing (LBAP), an alternative method for conduction system pacing. LBAP involves advancing a lead just beyond the tricuspid valve, positioned perpendicular to the basal right interventricular septum and then vigorously advanced into the septum aiming to penetrate the myocardium to reach the left bundle branch (see fi gures A and B). The landing site is larger than the His region allowing for a simpler procedure.
The resultant lead is position is stable and lead parameters exhibit excellent long term sensing and low capture threshold. Furthermore, follow up is no different to a standard pacemaker and physiological pacing is effectively achieved. I excitedly returned home only to suffer the worst “plane fl u” I had ever experienced which almost knocked me off my feet for the next 2 weeks. The world has since changed, but at the time, I took some OTC meds, avoided intubation and “soldiered on”.

Figure A. The His bundle is a small and discrete area. Positioning a lead at this location can be difficult due to instability, risk of dislodgement and increasing long term capture threshold. As it lies in a position between the atrium and the ventricle, there are often issues with over- or under sensing which may compromise device function. The LBB landing area is larger, as the left bundle branch fans out after its bifurcation beyond the distal His bundle deep within the septum. Consequently, it is easier to locate and lead stability and long-term parameters are superior to His bundle pacing.
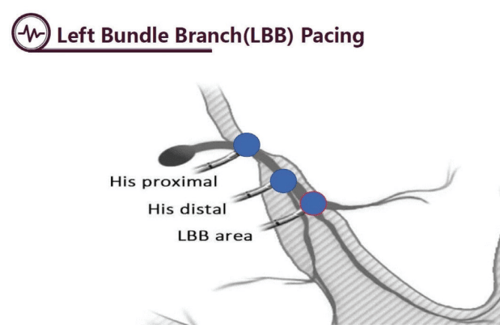
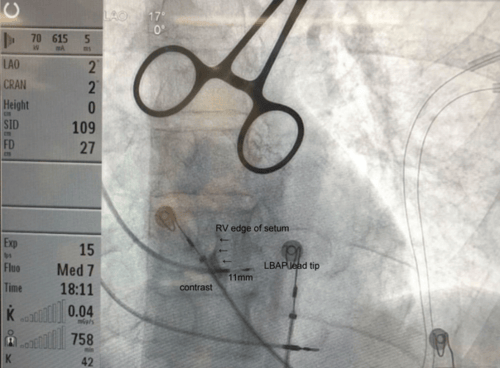
Figure B. This LAO image shows the final tip position of the LBAP lead within the interventricular septum. Note that contrast is being injected into the long sheath which has delivered the lead to the septum. The contrast delineates the right side of the interventricular septum and clearly demonstrates that the lead has penetrated the septum towards the left bundle branch. The distance between the lead helix tip to the proximal electrode is 11mm. The lead shown at the RV apex was later repositioned to the right atrial appendage and acts as back up pacing during the procedure.
Case Study:*
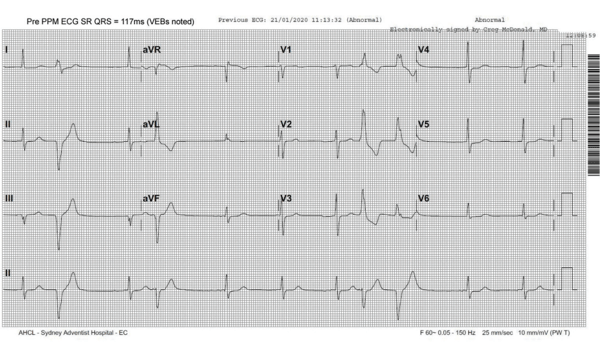
Mr RS is an 84yo man with idiopathic dilated cardiomyopathy, paroxysmal atrial fibrillation, and bradycardia.
His baseline ECG shows SR (with VEBs). Note the intrinsic narrow QRS width of 117ms

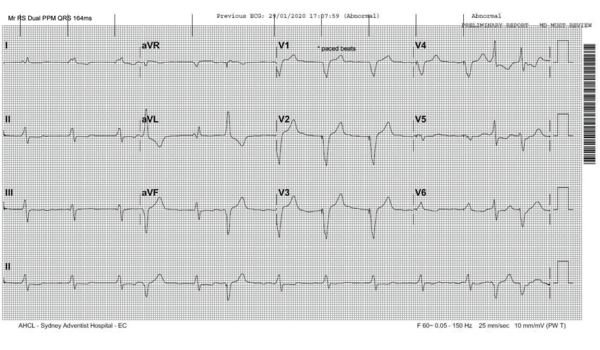
A Biotronik dual chamber pacemaker was implanted in January 2020 for symptomatic bradycardia. The post pacemaker ECG shows the RV paced beats have a QRS width of 164ms (the lead was positioned at the mid RV septum).
He subsequently developed heart failure with NYHA class III symptoms. Echocardiography revealed severe LV dysfunction with ejection fraction 32% and increased LV diastolic diameter of 6.2cm. In November 2020, he was referred for CRT defibrillator upgrade of his existing pacemaker in an attempt to improve LV function and as primary prophylaxis for ventricular arrhythmias. The new RV defibrillation lead was positioned at RV apex and the existing RV pacing lead was removed. Coronary sinus venography showed three potential targets, but two were not suitable (corkscrew origin and short vessel with distal tapering). A quadripolar LV lead was placed within the remaining anterolateral branch. However, phrenic nerve stimulation was noted when pacing poles 2, 3 and 4 but none when pacing pole 1 to RV coil. The threshold was excellent, and the lead position was accepted as there were no alternative targets.
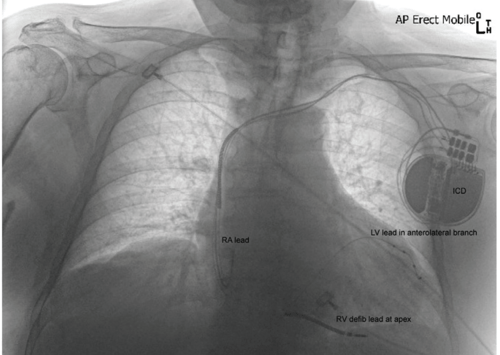
In December 2020 at the first ICD check, he reported recurrent intolerable phrenic nerve stimulation which could not be eliminated by programming. The LV lead was deactivated. In January 2021 he proceeded to LBAP lead placement. The LV lead had not dislodged which may have otherwise caused phrenic stimulation and was subsequently extracted. The LBAP lead was positioned successfully.
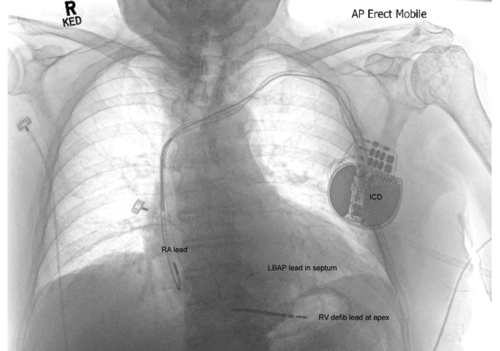
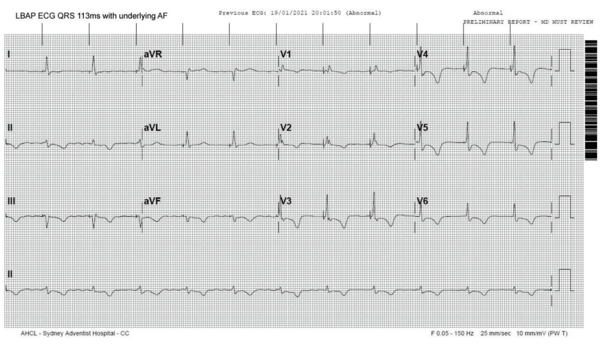
The ECG after LBAP upgrade shows underlying atrial fibrillation with narrow paced QRS complexes measuring 113ms. The morphology is similar to the intrinsic QRS before pacing (note the rSR’ pattern in V1).
At the February 2021 ICD check, LBAP lead parameters were stable and the patient reported subjectively improved symptoms. At the October 2021 ICD check, LBAP threshold was 0.8mV. He felt subjectively much improved and echocardiography showed a significant objective improvement with LV ejection fraction 45% and a reduced LV diastolic diameter of 5.7cm.
Discussion
As exemplified in the case above, effective resynchronisation can be achieved using physiological pacing. The pacing system is inherently simpler as it requires a single ventricular lead positioned at the left bundle area rather than a separate LV lead in the coronary sinus. Ironically, the most frequently used lead today for His bundle and LBA pacing is the very same lead used by my supervising professor in London almost 20 years ago, so the technology has been tried and tested. LBAP offers no disadvantage to pacing follow up, the pacing systems are technically MRI compatible and the implant procedure is relatively straight forward.
Conclusion
Despite hundreds of studies aiming to improve the responder rate for CRT using complex and time consuming echocardiographic measurements in years gone by, none were proven to be significantly better in predicting CRT response compared to a narrow paced QRS after device implant. CRT rarely results in a paced QRS morphology as close to normal as does physiological pacing. Early emerging data suggests that physiological pacing is superior to CRT with respect to improved LV function and that LBAP is not inferior to His bundle pacing in that regard. By preventing electrical dyssynchrony, physiological pacing will also likely eliminate the occurrence of pacemaker induced cardiomyopathy. Watch this space – I’m excited!
